|
From all outward appearances, they will look identical however. For example, both molecules with 2 bonds and 0 lone pairs and 2 bonds and 3 lone pairs will be linear. They will have different numbers of bonds and lone pairs, but the end result will be the same. One brief thing to take note of is that there are multiple linear and bent geometries. You'll see that the chart follows a very straightforward pattern of logic. The molecular geometry chart may seem like a lot of information at first, but over time it will become intuitive. The molecular geometry for a water molecule is bent, which is why the water molecule isn't a straight H-O-H molecule. If we look up at the table, we can find on the left side a column for molecules with 4 electrons groups, and within that group a molecule with 2 lone pairs. The central `O` atom has 4 groups of electrons around it: 2 bonds and 2 unpaired electrons, also called lone pairs. Let's say you had the formula of water, `H_2O`, and wanted to know its molecular geometry. In other words, by drawing out the Lewis structure of a molecule, one can determine the molecule's 3d orientation. We're going to discuss each one individually, but note that you can determine the molecular geometry of a molecule solely by the number of bonds and lone pairs around the central atom. The following chart provides the different molecular geometries and the conditions in which they arise. This will become important when discussing bond angles. 
If you substitute a single bond with a lone pair, the lone pair will repel the other electron groups away from it more than the single bond would. One key point is that lone pairs take up more room than single bonds do. The lone pair is usually not shown in 3d models and thus you have to visualize it yourself. This is why `NH_3` has a tri-pod shape: the lone pair on top of `N` pushes the other electrons (in the bond) away from it. The basic idea behind VSEPR is that electron pairs will repel each other. The only difference is the number of electrons on the central atom: `N` has a lone pair whereas `B` does not. Take a look at the Lewis structures of `NH_3` and `BF_3`. Molecular geometry is determined by the arrangement of valence electrons. The reason behind this is that the two structures have a different number and arrangement of valence electrons. This is the case of `BF_3` and `NH_3`: both have the general formula `AB_3`, but `NH_3` on the left is a tri-pod shape whereas `BF_3` on the right is a 3-legged starfish shape. In other words, two molecules with the general formulas `AB_3` may look completely different in real life: one may be a pyramid whereas the other may be completely flat. You have signed an examinee agreement, and it will be enforced on this subreddit.ĭo not intentionally advertise paid or free products or services of any sort.Valence Shell Electron Pair Repulsion (VSEPR) is a theory that states that the 3d orientation, also known as the molecular geometry, of a molecule is not dependent on its chemical formula but on the repulsion of valence electrons. 
We have one "stickied" post for each exam and score release day, contain all test day discussion/reactions to that thread only.ĭo not discuss any specific information from your actual MCAT exam. For an example format for submitting pictures of questions from practice material click hereĭo not link to content that infringes on copyright laws (MCAT torrents, third party resources, etc).ĭo not post repeat "GOOD LUCK", "TEST SCORE", or test reaction posts. These are considered spoilers and should be marked as such. Be nice to each other, hating on other users won't help you get extra points on the MCAT, so why do it?ĭo not post any question information from any resource in the title of your post. Rudeness or trolling will not be tolerated. Please message the moderators with your skills/ideas! MCAT RESOURCES & INFO Study Groups Want to help us improve this subreddit or tell us about a new resource we can add to the sidebar? Below you will find our forum rules, resources, and more. We request that you read the sidebar COMPLETELY before you post. r/MCAT is a place for support, discussion, advice, social networking, news, study tips and more. 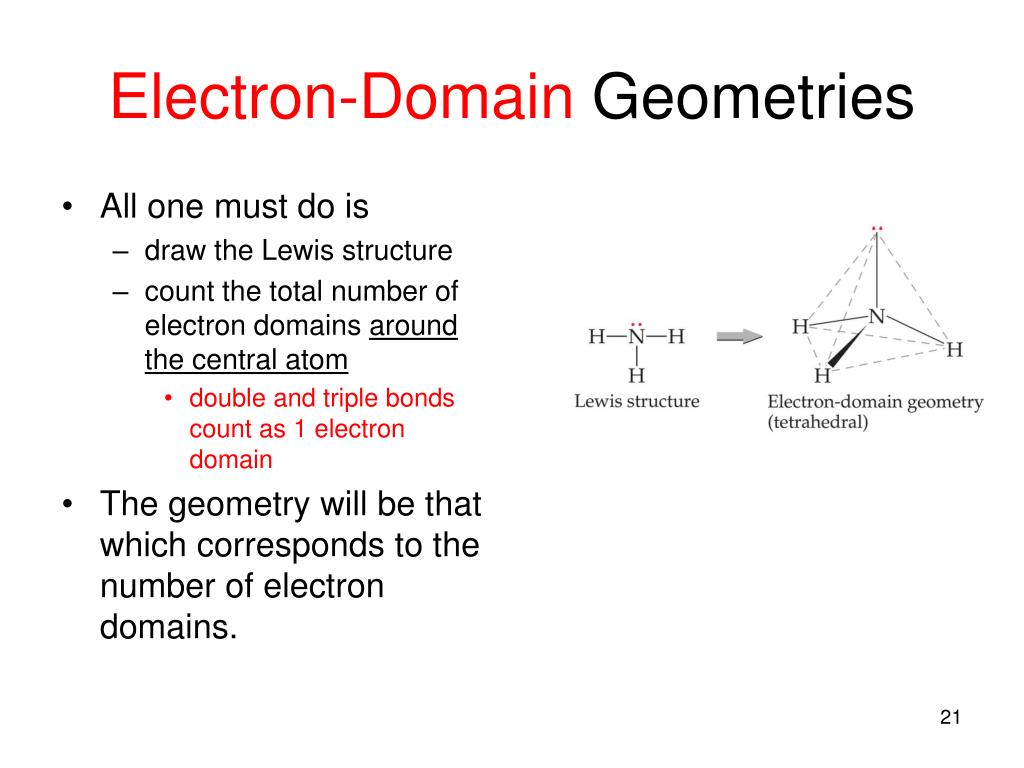 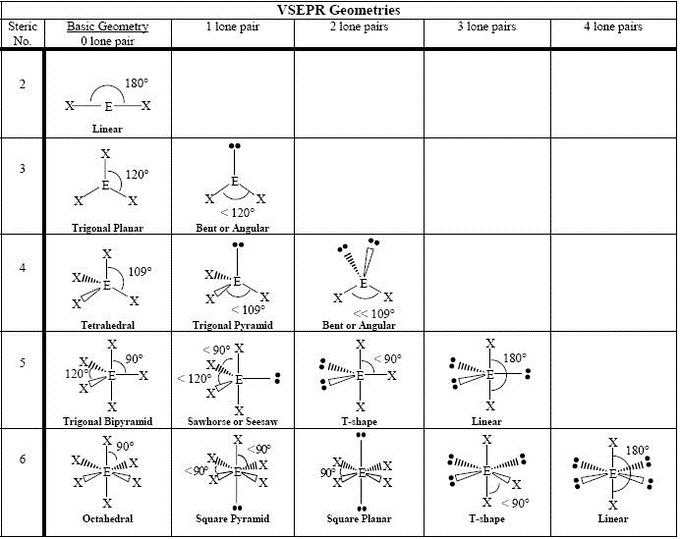
The MCAT (Medical College Admission Test) is offered by the AAMC and is a required exam for admission to medical schools in the USA and Canada. Welcome to the BEST place for MCAT prep and practice materials.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
